Prevalence, Diagnostic Utility and Associated Characteristics of Bronchodilator Responsiveness
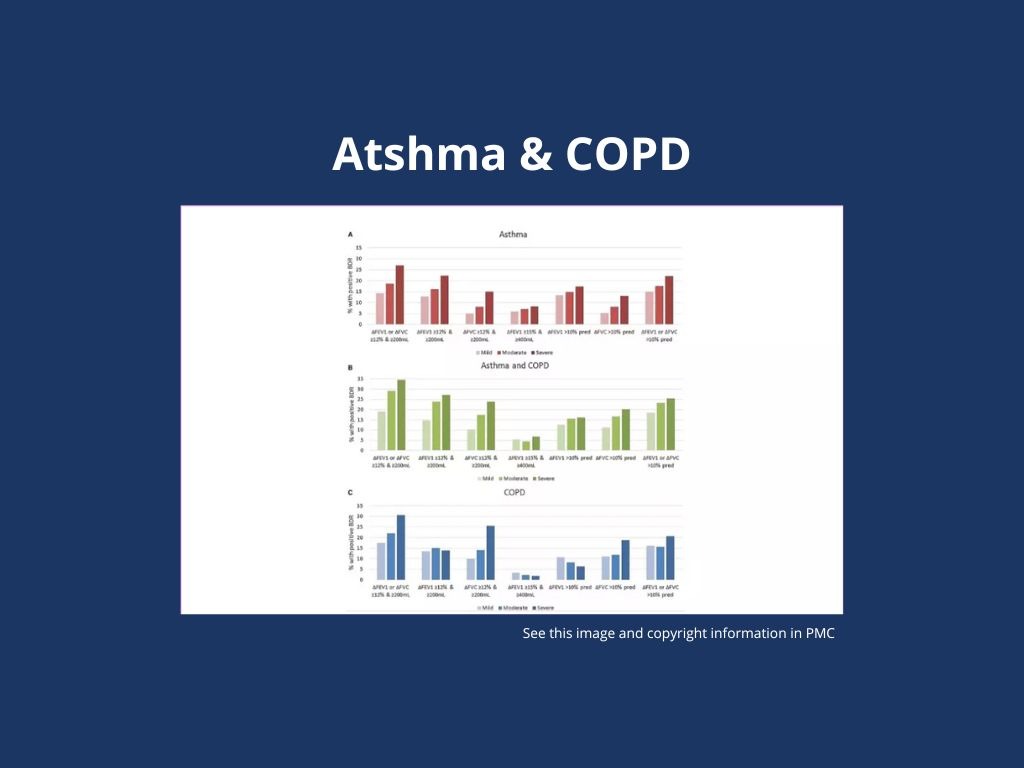
Bronchodilator Responsiveness (BDR) prevalence in patients with chronic airway diseases receiving treatment ranges from 18% to 30%, being modestly lower with the 2021 than with the 2005 European Respiratory Society/American Thoracic Society criteria, and it is associated with lower lung function and greater symptom burden.
These observations question the validity of BDR as a key diagnostic tool for asthma managed in clinical practice or as a standard inclusion criterion for clinical trials of asthma and instead suggest that BDR be considered a treatable trait for chronic airway disease.
Measurements and Main Results
3,519 patients were studied with a physician-assigned diagnosis of asthma, 833 with a diagnosis of asthma + COPD, and 2,436 with a diagnosis of COPD.
The prevalence of BDR was 19.7% (asthma), 29.6% (asthma + COPD), and 24.7% (COPD) using 2005 criteria and 18.1%, 23.3%, and 18.0%, respectively, using 2021 criteria.
Using 2021 criteria in patients diagnosed with asthma, BDR was associated with higher fractional exhaled nitric oxide; lower lung function; higher symptom burden; more frequent hospital admissions; and greater use of triple therapy, oral corticosteroids, or biologics. In patients diagnosed with COPD, BDR (2021) was associated with lower lung function and higher symptom burden.
Puedes leer el artículo completo aquí: https://pubmed.ncbi.nlm.nih.gov/38029294/
Autores: Richard Beasley, Rod Hughes, Alvar Agusti, Peter Calverley, Bradley Chipps, Ricardo Del Olmo, Alberto Papi, David Price, Helen Reddel, Hana Müllerová, Eleni Rapsomaniki.
Noticias relacionadas
A Brief History of COPD: As Told by Some of Its Senior Scientists and Clinicians
Explore the history of COPD and the evolution of disease concepts, treatment strategies, patient outcomes, pulmonary rehabilitation and clinical management.

Climate change and respiratory health: implications for respiratory clinicians in primary and specialist care
How climate change affects asthma and COPD, the role of air pollution and inhalers, and why sustainable respiratory care matters for patients and clinicians.

Indoor radon exposure and COPD in never smokers: a case–control study in a radon-prone area in Spain
Study on indoor radon exposure and its association with COPD risk and severity in never smokers in a radon-prone region.

A Brief History of COPD: As Told by Some of Its Senior Scientists and Clinicians
Explore the history of COPD and the evolution of disease concepts, treatment strategies, patient outcomes, pulmonary rehabilitation and clinical management.

Academia Europaea’s guidelines for the visualization of clinical outcomes
Academia Europaea proposes ring diagrams to improve clinical outcome visualization and support evidence-based healthcare decision-making.

Video del evento: Transformando la EPOC, claves para un manejo proactivo
Conferencia sobre el manejo proactivo de la EPOC centrada en el infradiagnóstico, el control clínico y el seguimiento estructurado del paciente.
Artículos
COPD
- 759578·Alberto Papi et Al.-Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY
- 759785·Richard Beasley et Al – Prevalence, Diagnostic Utility and Associated Characteristics of Bronchodilator Responsiveness
- 759788·Alvar Agustí, Rod Hughes, Eleni Rapsomaki, Barry Make, Ricardo Del Olmo, Alberto Papi, David Price, Laura Benton, Stefan Franzen, Jørgen Vestbo, Hana Mullerova – The many faces of COPD in real life: a longitudinal analysis of the NOVELTY cohort
- 759883·Alberto Papi, Rosa Faner, Ian Pavord, Federico Baraldi, Vanessa M McDonald, Mike Thomas, Marc Miravitlles, Nicholas Roche, Alvar Agustí. – From treatable traits to GETomics in airway disease: moving towards clinical practice
- 768799·Surya P Bhatt Richard Casaburi Alvar Agusti et Al. Chronic obstructive pulmonary disease: hiding in plain sight, a Statement from the COPD Foundation Medical and Scientific Advisory Committee
Imagen obtenida en artículo origninal.
